BACKGROUND: Recent World Health Organization (WHO) classifications (5 th edition), International Consensus Classification (ICC) recognized TP53-mutated ( TP53mut) myeloid neoplasms (MNs) as a distinct subcategory, acknowledging their uniformly poor outcomes. However, there are critical differences between the two guidelines in how TP53 allelic status is assigned. Both WHO-5 and ICC acknowledged the poor outcome of multi-hit TP53mut but excluded single-hit TP53mut. These changes are driven by a pivotal study demonstrating favourable outcome of single-hit TP53mut, however this study predominantly included de novo myelodysplastic syndromes (MDS) cases (Bernard et al. Nat Med 2020). In contrast, we previously reported poor outcome of TP53mut therapy-related myeloid neoplasm (t-MN) irrespective of the allelic status of TP53 (Hiwase et al Blood 2022andShah et al BCJ 2023). Finally, the ICC removed the subcategory of “therapy-related,” substituting it with diagnostic qualifiers instead. The WHO has grouped t-MN with secondary MN and renamed it as “myeloid neoplasm post cytotoxic therapy,” with the assertion that only multi-hit T P53mut MDS occurring post-cytotoxic therapy have poor outcome compared with single-hit.
METHODS: We analyzed one of the largest international cohorts of TP53mut MN (n=544) with full characterization of TP53mut allele status utilizing uniform ICC criteria and similar large numbers of de novo and t-MN cases managed at Mayo Clinic Enterprise (USA) and South Australian MDS Registry, Adelaide (Australia) with clinical, integrated genomic profiling, and long-term follow-up.
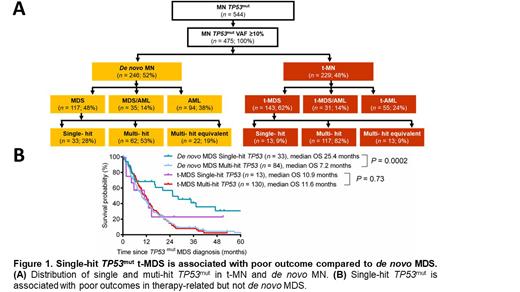
RESULTS: In our cohort of TP53mut MN (n=544), 475 (87.3%) MN had TP53mut variant allele frequency (VAF) of ≥10% and thus satisfied ICC criteria for TP53mut MN. Majority of TP53mut t-MN presented with MDS (bone marrow (BM) blasts 0-9%) compared de novo MN (62% vs. 48%; P=0.0012). While acute myeloid leukemia (AML) (BM blasts ≥20%) were more prevalent in TP53mutde novo MN compared to t-MN (38% vs. 24%; P= 0.001) and there was no difference in frequency of MDS/AML (BM blasts 10-19%) between the two cohorts (14% vs. 14%; P=0.8) ( Figure 1A).
Majority of TP53mut were observed in the DNA-binding domain in t-MN and de novo MN (95% vs. 96.2%; P=0.48). However, mutations in Tetramer Domain were more frequent in t-MN (4.3% vs. 0.3%; P=0.002), whereas those in the TAD2 domain were more frequent in de novo TP53mut MN (2.4 vs. 0%; P =0.006).
Integrating the mutation, conventional G-banding cytogenetic, copy number variation (CNV), FISH, and SNP array analysis, TP53mut MDS (BM blast 0-9%; n=260) were classified as multi-hit (n=179, 68.9%) or multi-equivalent (n=35, 13.5%) and single-hit (n=46, 17.7%) (Arber et al. Blood 2022). Striking enrichment of multi-hit/multi-hit equivalent was observed in TP53mut t-MDS compared to de novo MDS (91% vs. 72%; P<0.0001). While single-hit TP53mut were prevalent in de novo MDS compared to t-MDS (28% vs. 9%; P<0.0001). Importantly, median overall survival (OS) of single-hit was significantly longer than multi-hit de novo MDS (25.4 vs. 7.2 months; P=0.0002). In contrast to this we did not observe OS difference between single-hit and multi-hit TP53mut t-MDS (11.6 vs. 10.9 months; P =0.73) ( Figure 1B).
CONCLUSION: Our study based on a large international cohort of T53mut MN provides compelling evidence that in contrast to de novo MDS, single hit TP53mut t-MDS is associated with poor outcome. Our data supports the consideration of single hit t-MDS as a multi-hit equivalent.
Disclosures
Yeung:BMS: Research Funding; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Pfizer: Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Ross:Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Keros: Consultancy; Menarini: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees; Imago BioSciences, Inc., a subsidiary of Merck & Co., Inc., Rahway, NJ, USA: Research Funding; Celgene/BMS: Honoraria, Research Funding. Alkhateeb:Mayo Clinic: Current Employment. Foran:BeiGene: Membership on an entity's Board of Directors or advisory committees; BMS: Membership on an entity's Board of Directors or advisory committees; Actinium: Research Funding; Kura: Research Funding; Sellas: Research Funding; Roivant: Research Funding; Novartis: Research Funding; Celgene: Research Funding; Astellas: Research Funding; NCI: Membership on an entity's Board of Directors or advisory committees; CTI: Membership on an entity's Board of Directors or advisory committees. Patnaik:StemLine: Research Funding; Kura Oncology: Research Funding; CTI Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees. He:Kura Oncology, Inc: Consultancy. Shah:AbbVie: Research Funding; Astellas: Research Funding; MRKR Therapeutics: Research Funding; Celgene: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal